
Salt or cocrystal of salt? Probing the nature of multicomponent crystal forms with infrared spectroscopy - ScienceDirect

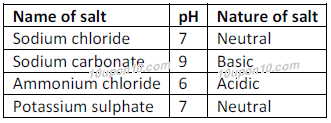
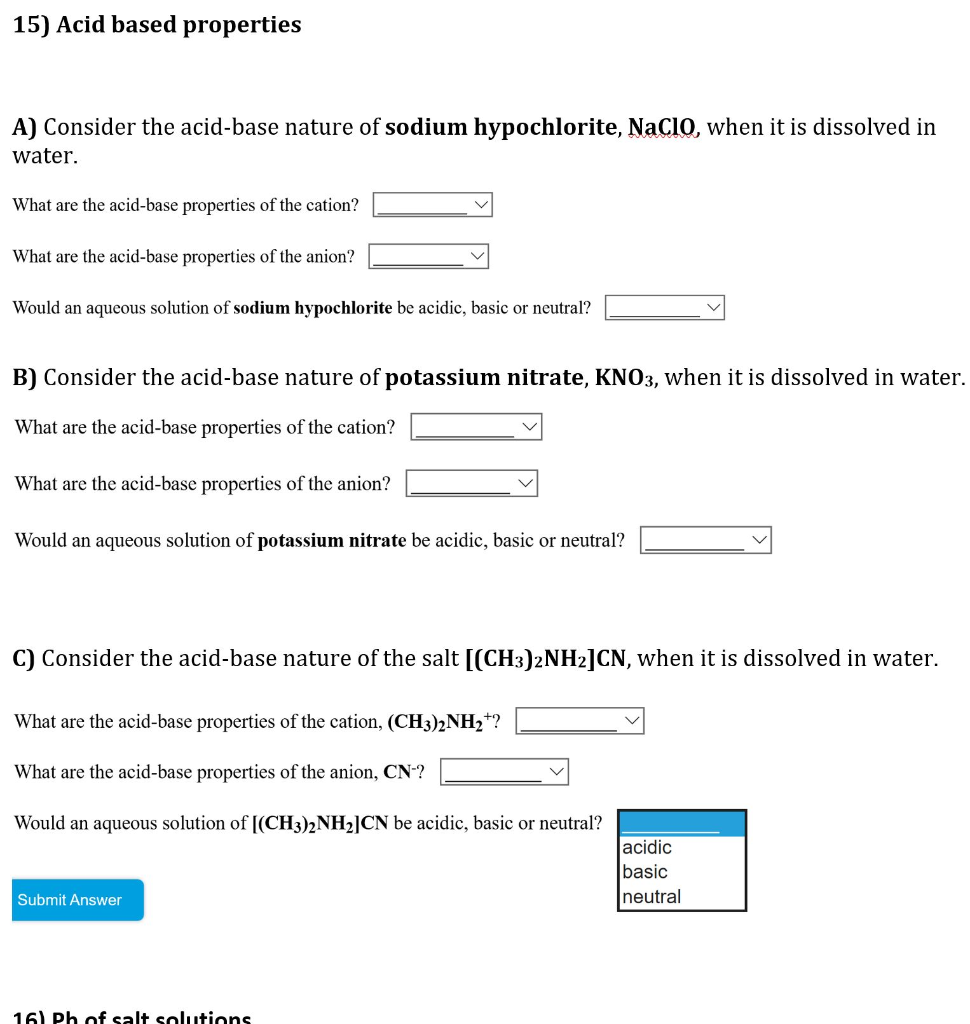
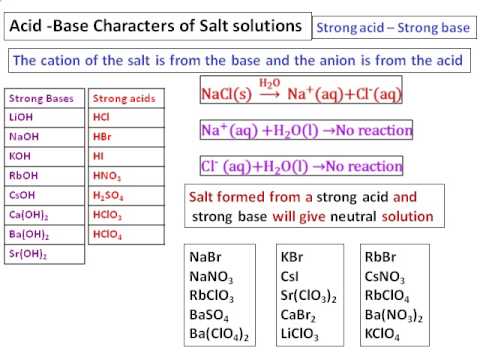
Identify the acid and base for ammonium chloride salt. What would be the nature of this salt? Mention - Brainly.in

Identity the acid and base for ammonium chloride salt. state the nature of this salt and mention its - Brainly.in







/salt-on-the-plains-of-salar-de-uyuni--bolivia--984096964-5c61dd4546e0fb00017dd6cd.jpg)











/__opt__aboutcom__coeus__resources__content_migration__mnn__images__2014__04__TypesOfSalt_m_0409-06e3f85e23464c748fd3108fbce6141d.jpg)