Vir Biotechnology to Provide Corporate Update and Report Third Quarter 2022 Financial Results on November 3, 2022
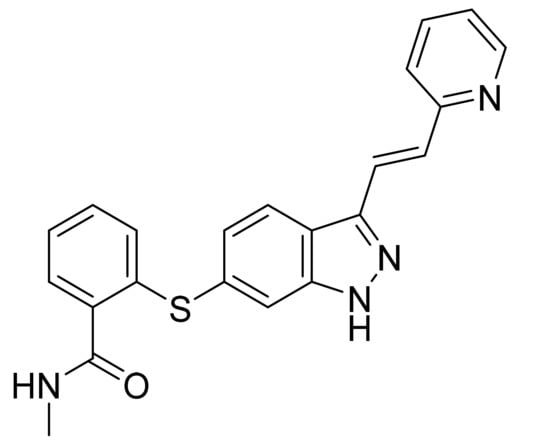
Pharmaceuticals | Free Full-Text | Innovations and Patent Trends in the Development of USFDA Approved Protein Kinase Inhibitors in the Last Two Decades | HTML

After making $1M+ on illegal trades, former Puma Biotechnology executive gets prison term | Fierce Pharma

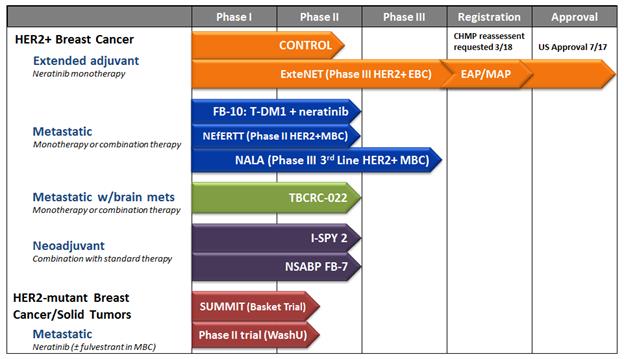
PUMA BIOTECHNOLOGY: Announces U.S. FDA Acceptance of Supplemental New Drug Application for Neratinib to Treat HER2-Positive Metastatic Breast Cancer | FDA Health News
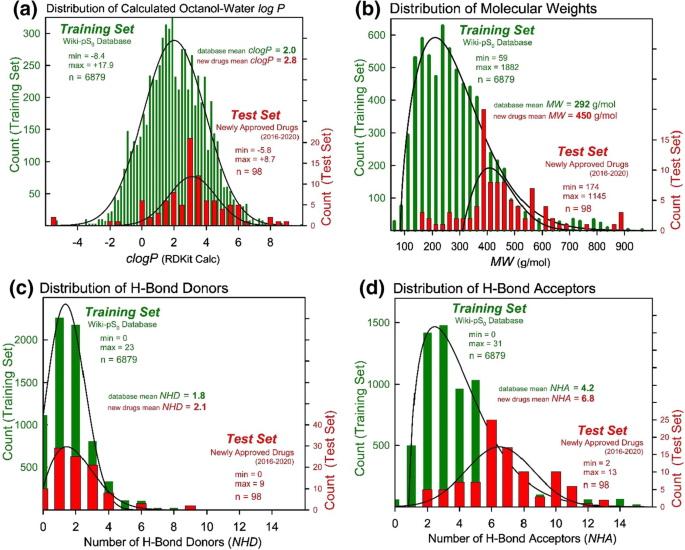
Predicting Solubility of Newly-Approved Drugs (2016–2020) with a Simple ABSOLV and GSE(Flexible-Acceptor) Consensus Model Outperforming Random Forest Regression | SpringerLink

Cancer hopeful neratinib plagued by diarrhea, analysis shows, but Puma cites a solution | Fierce Pharma